 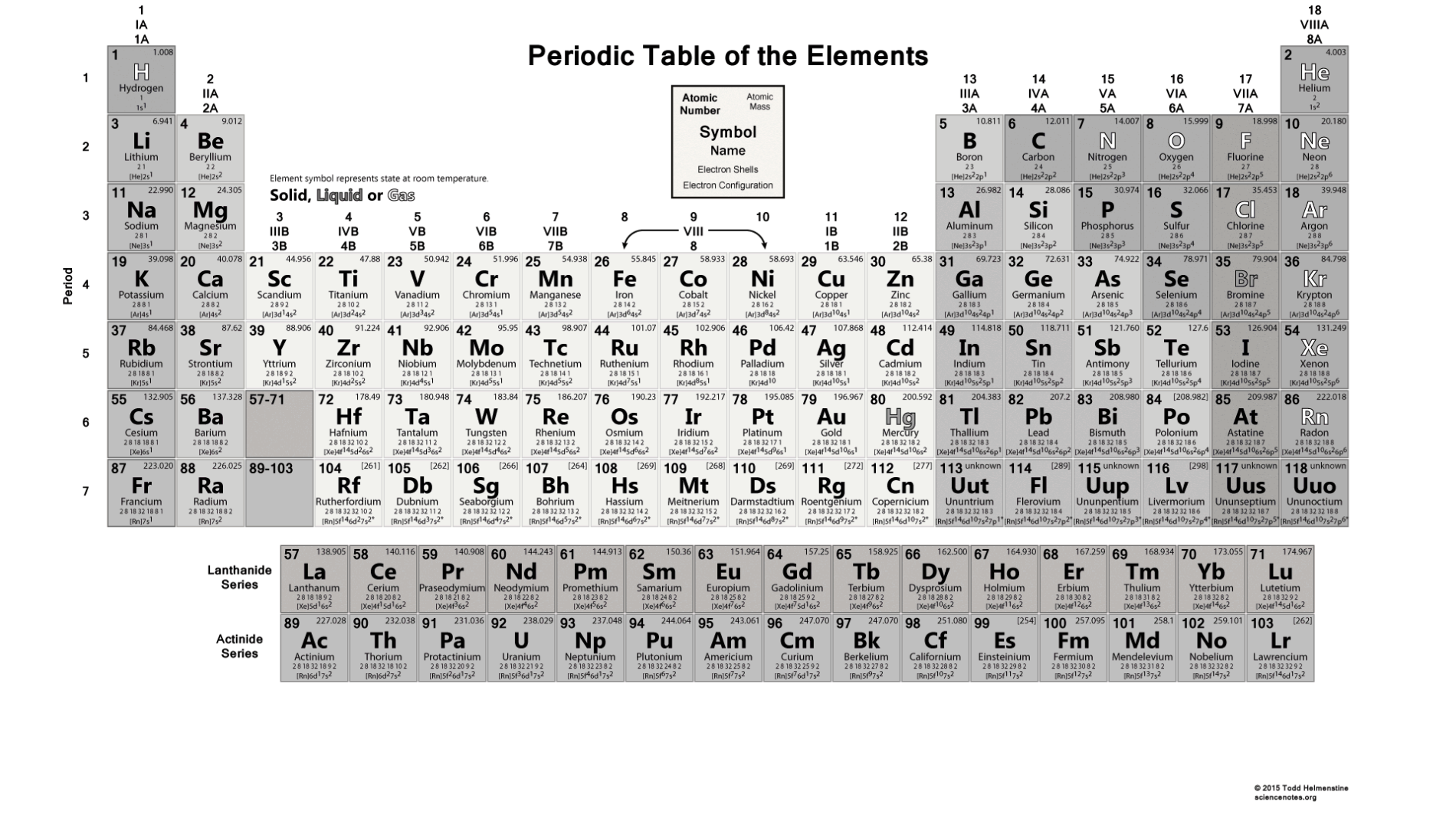
It is a tabular arrangement of the elements by their chemical properties that usually uses abbreviated chemical symbols in place of full element names, but the linear list format presented here is also useful. The definitive visualisation of all 118 elements is the periodic table of the elements, whose history along the principles of the periodic law was one of the founding developments of modern chemistry. A chemical element, often simply called an element, is a type of atom which has the same number of protons in its atomic nucleus (i.e., the same atomic number, or Z). This is a list of the 118 chemical elements that have been identified as of 2023. Namely, isotope, isobar, and isotone.List of the 118 identified chemical elements Therefore, the number of neutrons in carbon is 6.īased on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered. We know that the atomic number of carbon is 6 and the atomic mass number is 12. That is, neutron number (n) = atomic mass number (A) – atomic number (Z) Thus, the number of neutrons in an element is obtained from the difference between the number of atomic masses and the number of atoms. That is, the number of atomic masses (A) = p + n 
Therefore, the total number of protons and neutrons is called the atomic mass number. Therefore, atomic mass refers to the total mass of protons and neutrons.Ītomic mass(A) = Nucleus mass = Total mass of protons and neutrons (p + n)Īgain, the mass of each proton and neutron is about 1amu. The nucleus is made up of protons and neutrons. Therefore, the mass of the nucleus is called atomic mass. One is a positively charged particle proton and the other is a charge-neutral particle neutron.Īlmost all the mass of the atom is accumulated in the nucleus. There are two types of particles in the nucleus. We already know that the nucleus is at the center of the atom. Relationship between the atomic mass and carbon atomic number This site has published an article detailing the electron configuration of carbon that you can read if you want. To know these properties of carbon one must know the atomic number of carbon. Therefore, carbon is the p-block element. The last electron of carbon enters the p-orbital. So, the valence electrons of carbon are four. And the last shell has a total of four electrons. The last shell of carbon has four unpaired electrons, so the valency of carbon is 4. The properties of an element can be determined by electron configuration. The atomic number can be used to determine the number of electrons in an element and the exact position of an element in a periodic table. Therefore, the number of negatively charged electrons orbiting in its orbit is equal to the number of positively charged protons in the nucleus.Ītomic number (Z) = Number of charges in the nucleus (p) Importance of the atomic number of carbonĪn atomic number is a number that carries the properties of an element. That is the total number of protons in the atomic number. We know that protons are located in the nucleus of an atom as a positive charge. This number is equal to the serial number of the periodic table. The atomic number of the element is expressed by ‘Z’. Thus, the number of positive charges present in the nucleus of an element is called the atomic number of that element. He named that number the order of the atoms. The results of the experiment show that each element has a unique integer equal to the number of positive charges in the nucleus of that element. Scientist Henry Gwyn Jeffreys Moseley researched the X-ray spectrum of various elements from 1913 to 1914. Carbon is a p-block element and its symbol is ‘C’. Therefore, the atomic number of carbon(C) is 6. The carbon atom contains a total of six electrons and protons. The sixth element in the periodic table is carbon. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
